Abstract
Introduction
The first two trials to test the activity of daratumumab (DARA) in relapsed/refractory multiple myeloma (MM) were GEN501 (DARA monotherapy) and GEN503 (DARA in combination with lenalidomide [LEN] and dexamethasone [DEX]). GEN501 enrolled 104 participants from 2008 and GEN503 enrolled 45 participants from 2012. GEN501 has been completed; GEN503 is active but has finished accrual. We are reporting on ten patients that are still responding to DARA therapy, several years after their inclusion in the GEN501 and GEN503 trials.
Methods
Ten patients were included from three participating centers in Denmark, The Netherlands and Sweden. DARA therapy had to be continuous from the initiation of trial participation with no evidence of progressive disease. Data were collected retrospectively from patient charts. Clinical characteristics, ISS and FISH results were assessed at the time of diagnosis. Lines of myeloma therapy prior to inclusion in GEN501 or GEN503 were reviewed; duration of therapy, time to next therapy (TNT) and response to each of the prior lines of therapy were determined. Results of minimal residual disease (MRD) assessments were reviewed.
Results
Ten patients (six men, four women) were identified, three from GEN501 and seven from the GEN503. Four out of seven patients in GEN503 stopped LEN and DEX treatment due to side effects, but all ten patients are still treated with DARA intravenously every four weeks.
Patients were diagnosed with MM between 2005 and 2012. The median age at diagnosis was 59 years (range 49-62). Seven patients had IgG-kappa, two had kappa light-chain only and one patient had IgG-lambda M-protein. Nine patients had anemia, eight had bone disease, three had renal insufficiency and one had hypercalcemia at diagnosis. ISS was available in nine patients. Three patients had ISS I, five had ISS II and one had ISS III. FISH at diagnosis was available in eight patients; none of which had high-risk cytogenetics, defined as the presence of either del 17p, t(4;14) or t(14;16).
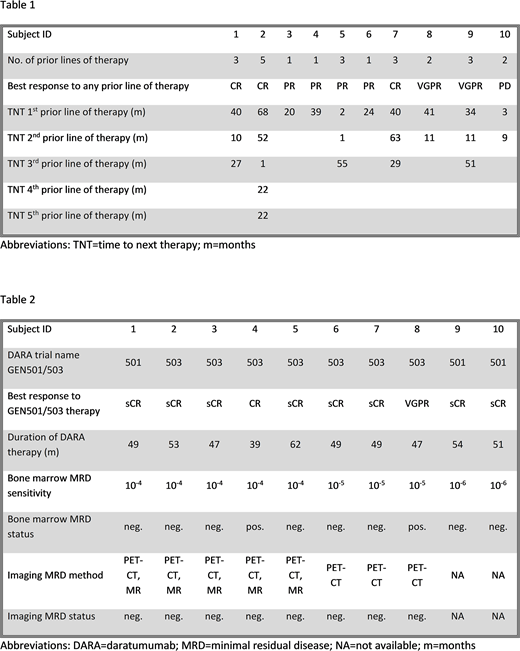
The prior lines of therapy are summarized in Table 1. The median number of prior lines of therapy was 3 (range 1-5). Nine patients had received high-dose melphalan with autologous stem cell transplantation; one of these was a tandem autologous transplantation. All patients had been treated with bortezomib; seven patients had been treated with LEN. Three patients were refractory to bortezomib; two were refractory to LEN. The best response to any prior line of therapy was CR in three, VGPR in two, PR in four patients and PD in one patient. The median TNT was 37 months (range 2-68) for the first (n=10), 11 months (range 1-63) for the second (n=7) and 29 months (range 1-55) for the third (n=5) prior line of therapy. One patient had been treated with five prior lines of therapy; the TNT for the fourth and fifth lines were 22 months each.
DARA therapy and MRD results are summarized in Table 2. Patients were enrolled in GEN501 or GEN503 between 2013 and 2015. The median time from diagnosis to the start of DARA therapy was 57 months (range 12-168). The best response to therapy was sCR in eight patients, CR in one patient and VGPR in one patient. The median duration of DARA therapy was 49 months (range 39-62). Eight of ten patients achieved a longer response to DARA therapy with or without LEN and DEX than to any previous regimen. Bone marrow MRD assessments were carried out by flow-cytometry between 2014 and 2018 in all patients. The sensitivity of the used assay was 10-6 in two, 10-5 in three and 10-4 in five patients. Eight subjects were bone marrow MRD-negative and two subjects were MRD-positive at a sensitivity of 10-4 and 10-5, respectively. Imaging MRD assessments were carried out in eight patients between 2017 and 2018. PET-CT was carried out in all eight patients; diffusion-weighted whole-body MRI was carried out in five patients. All patients were imaging MRD-negative.
Conclusions
Continuous DARA treatment, either as monotherapy or combined with LEN and DEX can achieve MRD-negative disease and unprecedented long-lasting remissions in select patients with relapsed or refractory multiple myeloma in the absence of high-risk cytogenetics.
Minnema:Amgen: Consultancy; Takeda: Consultancy; Celgene: Consultancy, Research Funding; Janssen: Consultancy; Servier: Consultancy. Plesner:Celgene: Other: Independent Response Assessment Comittee; Janssen: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal